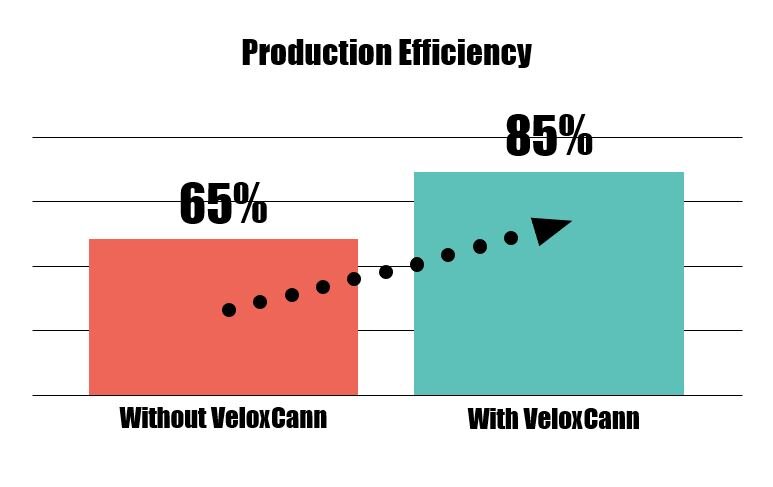
Increasing Production Efficiency by 20%
Meet Lisa
Lisa is Vice President of Operations at a young but rapidly growing, GMP-compliant Contract Development and Manufacturing Organization (CDMO). Her organization serves other companies in the pharmaceutical industry on contract basis to provide development and manufacturing of capsules and tablets at their GTA lab facility.
Tasked with managing business operations, ranging from cash flow to operational performance to human resources, Lisa understands that to ensure the growth and survival of the young company, product quality and speed to market must keep up with customer demand – otherwise, customers will go elsewhere and their reputation will be greatly impacted. Like any young and growing company, there was and remains a continuous challenge for Lisa to get everything done with a lean team.
The Challenge
The manufacturing environment is fast-paced and like any supervisor, Lisa was faced with multiple demands, all vying for her attention. These constantly changing demands forced her into firefighting mode, which is not an efficient long-term strategy. This reactive approach resulted in over 2,000 total hours of downtime per year – from full shut-downs for mandatory training to equipment breakdowns – and exposed the company to other detrimental risks such as product recalls.
“These constantly changing demands forced her into firefighting mode, which is not an efficient long-term strategy.”
Lisa wanted to reduce the number of fires but encountered hurdles ranging from time-constraint to not having the technical expertise to effectively implement the appropriate systems to benchmark, accurately document, and proactively monitor relevant KPI metrics (e.g. safety, quality, yield, and productivity) to pinpoint and troubleshoot problematic areas.
She also found maintaining GMP compliance standards and delivering training were both costly and time-consuming, in addition to the time and resources spent managing equipment, vendors, and other non-value added activities on an ad hoc basis that could otherwise be spent focusing on more strategic growth initiatives.
The Solution
For Lisa, success meant the company punching above their weight with its existing team and resources. She wanted to implement a more proactive system that would fit within the company’s means and values while empowering her team with the access to an on-site and real-time support structure of an established enterprise. More importantly, she wanted to level-up operations without disrupting either its “all-hands-on-deck” culture or advantages of being lean, nimble, and not burdened by a costly and rigid software system overhaul that may not be the right fit to deliver the optimal ROI.
“success meant the company punching above their weight with its existing team and resources.”
Lisa turned to VeloxCann for on-site help with: 1) improving operational efficiencies; 2) looking after their equipment; and 3) structuring their employee and development programs. VeloxCann’s system and training solutions have been refined over 30 years of pharmaceutical experience in a GMP environment to reduce downtimes and improve operations.
The Result
With an extra pair of eyes alongside her QA team to proactively monitor quality parameters by leveraging VeloxCann’s on-site service engineer and analytics management system along with a roster of more versatile and skilled workers developed from cross-training and knowledge-sharing to bridge skill gaps, Lisa’s team would accelerate product speed to market by improving their first pass yield by 10-30%, reducing approximately $200,000 a year in waste.
“Leveraging a roster of more versatile and skilled workers developed from cross-training and knowledge-sharing to bridge skill gaps.”
By mitigating both unforeseen employee absences and equipment maintenance, VeloxCann would help to reduce downtime by 40%, saving a total of about 900 hours annually. This creates extra capacity to produce ~40,000,000 more capsules per year. The VeloxCann program also helps Lisa to reduce 40-50 additional hours of downtime annually per employee for mandatory training (which are standalone hours that are not embedded in work activities) by coinciding carefully curated modules with planned scheduled maintenances, creating value even during “downtime”. The company’s employees now understand the impact and value of downtime and waste.
Reducing both waste and downtime boosts production efficiency. Ultimately, with VeloxCann by their side, Lisa and her team are able to stay steps ahead of fires to deliver quality product to market faster while also creating more time and resources to drive sales and R&D.